You navigate a world defined by invisible scientific triumphs every single day. When you swallow a pill to cure a sudden infection, undergo a routine diagnostic scan, or roll up your sleeve for a seasonal booster, you rely on a cumulative legacy of rigorous observation, fierce debate, and painstaking peer review. Tracking breakthroughs across physics, chemistry, space, and technology reveals a clear pattern—our most profound leaps in human health rarely happen in isolation. Understanding the ten foundational medical discoveries of human history provides you with a crucial framework for interpreting today’s rapid-fire biotech announcements. You need to grasp how early researchers validated their claims to truly appreciate the statistical confidence backing modern genomic therapies and artificial intelligence-driven drug designs.
Medical milestones do not spring forth fully formed; they demand years of meticulous experimentation, replication, and intense scrutiny from the global scientific community. From the earliest realizations that invisible organisms dictate disease, to the precise manipulation of your genetic source code, each breakthrough serves as a stepping stone. As you explore these ten monumental shifts in health science, you will recognize how historical data continues to inform the clinical trials, pharmacological standards, and surgical procedures that safeguard your life right now.

Breaking Hook: The Genesis of Modern Healing
You might take an everyday antibiotic prescription for granted today, but merely a century ago, a simple scratch from a rose bush could act as an absolute death sentence. The transformation of medicine from anecdotal guesswork to a rigorous, data-driven science began with the establishment of the germ theory of disease. Before the mid-nineteenth century, the prevailing medical consensus blamed illnesses on toxic air or miasma. Louis Pasteur shattered this paradigm in the 1860s through brilliantly simple methodologies. By boiling nutrient broth in swan-neck flasks—which allowed air to enter but trapped dust and microbes in the curve of the glass—Pasteur provided irrefutable empirical data disproving spontaneous generation. Robert Koch soon followed by identifying the specific microscopic bacteria responsible for anthrax, cholera, and tuberculosis. These milestones created the ultimate “who, where, and why” of infectious disease control, drastically shifting demographic survival rates globally.
Building upon the foundational understanding that pathogens cause illness, researchers turned their focus toward systemic prevention and targeted eradication. Edward Jenner had previously established the concept of vaccination in 1796 when he observed that milkmaids who contracted cowpox remained immune to the far deadlier smallpox. Jenner tested his hypothesis by intentionally inoculating an eight-year-old boy with cowpox—a method considered wildly unethical by your modern clinical standards, yet it initiated the global practice of immunization. This historical foundation paved the way for Alexander Fleming’s 1928 identification of penicillin. Fleming famously returned to a messy laboratory to find a stray mold, Penicillium notatum, halting the growth of staphylococci bacteria in a petri dish. While Fleming published his initial findings in the British Journal of Experimental Pathology, the true breakthrough in application required massive wartime funding. During World War II, researchers Howard Florey and Ernst Chain secured government grants to purify and mass-produce the compound, introducing an era of antibiotics that slashed infectious mortality rates and fundamentally altered global population dynamics.
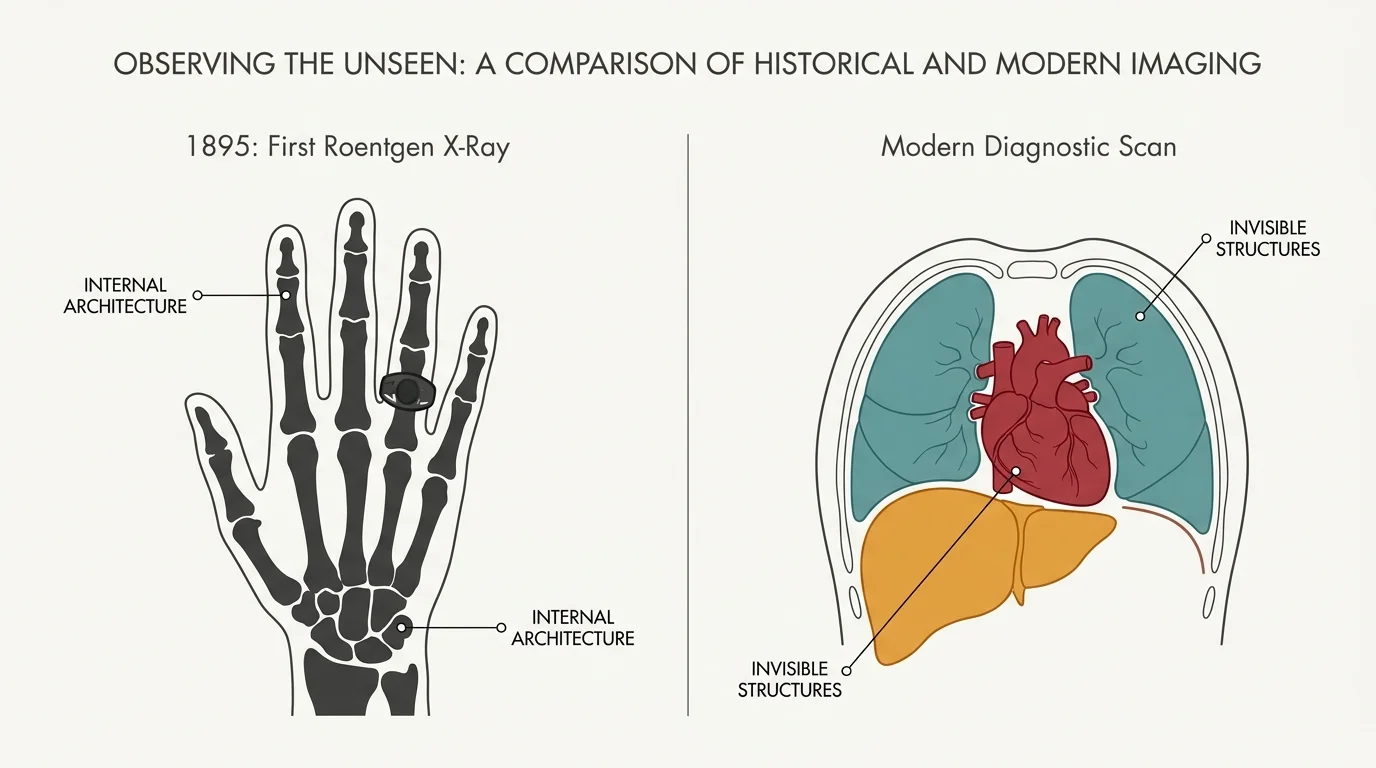
Method & Results: Observing the Unseen
When you undergo an invasive surgical procedure, your survival and comfort depend on a precise cocktail of chemical agents and a deep understanding of your invisible internal architecture. The discovery of modern anesthesia transformed the surgical theater from a place of agonizing, frantic trauma into a controlled environment designed for precise, methodical intervention. On October 16, 1846, William T.G. Morton conducted a public demonstration at Massachusetts General Hospital, successfully using inhaled diethyl ether to render a patient completely unconscious during the removal of a neck tumor. The observational methods utilized in early anesthesia trials relied heavily on monitoring patient responsiveness and respiratory rates, eventually yielding replicable protocols that allowed surgeons to operate deeper inside the human body with robust statistical confidence in patient survival.
Fifty years later, the ability to observe the human body took another quantum leap with Wilhelm Röntgen’s 1895 discovery of X-rays. While experimenting with cathode ray tubes in a darkened laboratory, Röntgen noticed a barium platinocyanide screen glowing brilliantly across the room. He systematically placed various objects between the tube and the screen, eventually capturing the first radiological image of his wife’s hand, clearly revealing her bones and wedding ring. Röntgen’s swift publication and the immediate global replication of his experiments highlight a rapid, transparent peer-review process that forever altered diagnostic medicine. Within months, field hospitals deployed early X-ray machines to locate shrapnel in wounded soldiers without resorting to exploratory surgery.
Simultaneously, scientists recognized that navigating the human body required an understanding of its unique chemical signatures. In 1901, Karl Landsteiner revolutionized trauma care by discovering the ABO blood group system. By methodically centrifuging blood samples, separating serum from red blood cells, and cross-matching them among his colleagues, Landsteiner recorded distinct patterns of agglutination—the clumping of incompatible cells. This concrete data set established the parameters necessary for safe blood transfusions, drastically reducing surgical mortality rates.
The quest to map the unseen culminated in 1953 when James Watson, Francis Crick, and Rosalind Franklin mapped the double-helix structure of deoxyribonucleic acid (DNA). Franklin’s legendary Photo 51—produced through incredibly precise X-ray crystallography methods—provided the undeniable physical evidence of the molecule’s helical nature. By calculating the exact molecular distances necessary to satisfy the hydrogen bonding between specific nucleotide bases, the team secured the physical blueprints of biological inheritance. When they published their concise findings in the journal Nature, they launched the modern era of molecular biology, allowing researchers to sequence the very source code of your existence.
Applications & Debate: Taming Chronic and Viral Threats
Managing a chronic illness or surviving a rapid viral epidemic profoundly shapes how you interact with the modern healthcare system. The discovery and isolation of insulin provides a masterclass in translating raw laboratory data into immediate human applications. In 1921, Frederick Banting and Charles Best conducted a series of canine experiments at the University of Toronto, proving that extracted pancreatic secretions could successfully regulate blood sugar levels. They swiftly moved to human applications, administering their purified extract to Leonard Thompson, a fourteen-year-old boy dying from diabetic ketoacidosis. The results were statistically staggering; the boy’s dangerously high blood glucose levels plummeted, pulling him out of a critical coma. However, the application of this life-saving discovery immediately sparked controversies over intellectual property and ethics. Banting and his colleagues sold their patent to the university for a single dollar to ensure universal public access; yet today, you watch pharmaceutical companies and international policymakers fiercely debate insulin pricing, patent evergreening, and market exclusivity.
Antiviral therapies offer another powerful example of how scientific discoveries navigate the turbulent waters of application, debate, and profound ethical tension. When researchers first identified the Human Immunodeficiency Virus (HIV) as the causative agent of AIDS in the early 1980s, the scientific community faced intense public pressure to deliver practical, life-saving therapies. Activist organizations fundamentally changed how clinical trials functioned by demanding faster access to experimental drugs like AZT. The ensuing clinical trials introduced accelerated regulatory approval pathways, drastically altering how agencies evaluate and distribute emergency medications. While early single-drug regimens faced challenges with viral mutation and resistance, the eventual introduction of highly active antiretroviral therapy (HAART) provided overwhelming statistical confidence in expanded survival curves. This breakthrough transformed a uniformly terminal diagnosis into a manageable chronic condition, though it raised ongoing global debates regarding patent waivers, intellectual property rights, and the equitable distribution of generic drugs in developing nations.

Global Context: The Geopolitics of Genomic Medicine
The tenth and most recent foundational discovery—CRISPR-Cas9 gene editing—brings all previous biological milestones into a fiercely competitive geopolitical arena. In 2012, researchers Jennifer Doudna and Emmanuelle Charpentier published a landmark paper wherein they demonstrated how to program a bacterial immune system to precisely slice and edit targeted DNA sequences. You are currently witnessing an unprecedented international race to commercialize and optimize this technology. The Broad Institute of MIT and Harvard engaged in a brutal, decade-long patent dispute with the University of California, highlighting the massive financial stakes and venture capital investments tied to genetic medicine.
Furthermore, the global context of CRISPR involves deep geopolitical tensions and constantly shifting ethical boundaries. When a biophysics researcher in China announced the creation of the world’s first gene-edited human twins in 2018, the global scientific community universally condemned the rogue experiment. Critics cited the total lack of standard peer review, the direct violation of international ethical norms, and the complete absence of institutional oversight regarding germline editing—alterations that can be passed down to future generations. Today, organizations like the World Health Organization collaborate with national regulatory bodies to establish universal guidelines for human genome editing. Major funding sources ranging from the United States Department of Energy to the National Institutes of Health aggressively back competing laboratories, ensuring that genetic engineering remains heavily intertwined with international economic supremacy and global health security.
FAQ: Navigating the Medical Milestone Landscape
How long does it typically take for a medical discovery to reach the public?
You can generally expect a timeline of ten to fifteen years from initial laboratory observation to widespread clinical availability. Researchers must shepherd experimental compounds through rigorous preclinical testing before advancing to Phase I, II, and III human clinical trials to establish safety profiles and statistical efficacy. Regulatory bodies mandate these extended timelines, requiring double-blind, placebo-controlled studies to prevent toxic side effects from reaching the general population.
Where can you access the raw data for modern clinical trials?
If you want to verify the claims made by pharmaceutical companies or academic research institutions, you should utilize comprehensive public registries. Platforms such as ClinicalTrials.gov host extensive, government-mandated data regarding study protocols, participant demographics, and primary outcomes. Additionally, many researchers now upload their initial findings to open-access preprint servers like medRxiv, allowing you to review the raw science before formal peer review concludes.
How do these historical milestones impact current pharmaceutical industry practices?
Every modern drug development pipeline relies directly on these ten foundational discoveries. When an artificial intelligence algorithm predicts how a newly synthesized cancer drug will bind to a specific cellular receptor, it relies entirely on the structural biology principles established by the discovery of DNA and protein architectures. You cannot separate today’s advanced synthetic biology and targeted immunotherapy from the basic tenets of germ theory, immunology, and observational methodology proven over a century ago.
How can you follow credible updates on emerging medical breakthroughs?
You should bypass sensationalized social media claims and look directly to primary scientific sources and established medical journals. Publications like The Lancet, Nature, and the New England Journal of Medicine maintain rigorous standards for peer review and methodological transparency. Following direct announcements from major public health agencies and international scientific panels ensures you receive practical, actionable insights free from aggressive commercial bias or misleading interpretations.
Forward Track: The Next Generation of Medical Milestones
As you look toward the horizon of human health, the convergence of these historical discoveries with modern computing power promises unprecedented medical milestones over the next decade. Researchers currently deploy advanced machine learning models to predict the three-dimensional structures of virtually all known biological proteins, accelerating targeted drug discovery by decades. You will soon see mRNA technology—validated on a massive global scale during recent pandemic vaccine rollouts—repurposed to target individualized cancer tumors, autoimmune disorders, and rare genetic deficiencies.
Space-based research also introduces a fascinating and highly practical frontier for human healing. The microgravity environment aboard the International Space Station—free from the convective forces and sedimentation found on Earth—allows scientists to grow larger, more perfectly formed protein crystals than those synthesized in terrestrial laboratories. This ongoing orbital research directly informs the precise molecular design of new monoclonal antibodies and targeted radiation therapies. As international space agencies and highly funded commercial partners launch the next generation of privately operated space stations, you can expect extraterrestrial manufacturing capabilities to yield novel treatments for the most stubborn terrestrial diseases. The scientific method that guided researchers over the past two centuries continues to rapidly evolve, ensuring that the next great medical discovery is already quietly forming in a laboratory, a supercomputer, or perhaps in low Earth orbit.













