Scientists now treat aging not as an inevitable decline but as a biological process you can measure, alter, and potentially slow down. Understanding the cellular mechanisms driving this wear and tear gives you actionable tools to protect your healthspan and extend your vital years. For decades, researchers assumed our bodies simply rusted like old machines over time. Modern science dismantled that belief by uncovering the specific genetic and metabolic pathways that dictate how we age. By identifying senescent cells, telomere degradation, and nutrient-sensing networks, longevity research shifted from science fiction into concrete medicine. These breakthroughs mean the timeline of your life is far more malleable than previously imagined.
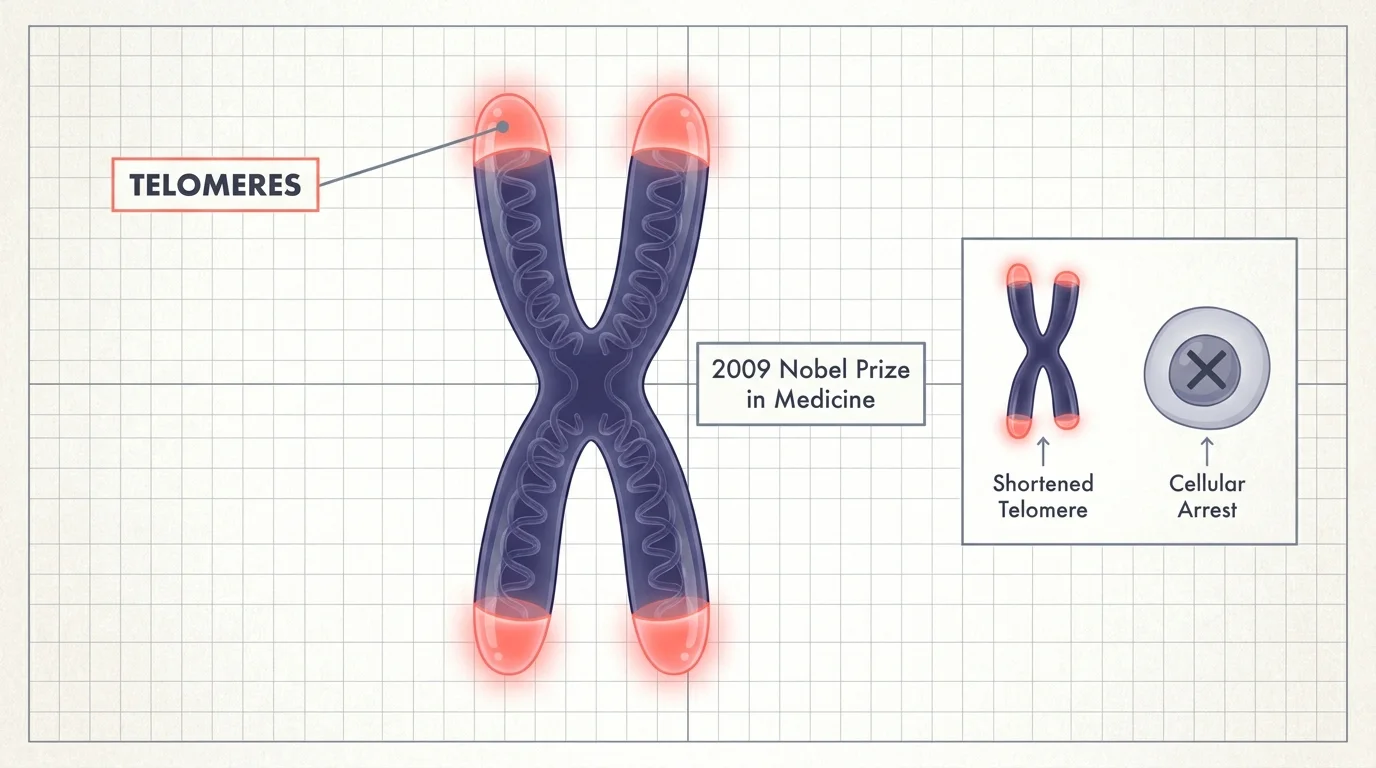
1. Telomeres and the Biological Countdown
Your cells have a built-in countdown clock known as telomeres, which act like the plastic tips on shoelaces protecting your DNA strands. Every time a cell divides, these protective caps shorten; once they become too brief, the cell stops dividing and tissue regeneration halts. Researchers awarded the 2009 Nobel Prize in Medicine discovered telomerase, an enzyme that can rebuild these caps. This revelation proved that cellular mortality has a distinct genetic trigger rather than occurring through random wear. You can directly influence your telomere attrition rate through lifestyle factors; chronic stress accelerates the shortening process, while regular cardiovascular exercise preserves telomere length. This paradigm shift established that your biological timeline relies heavily on your environment and habits.

2. Senescent Cells and Tissue Inflammation
As your body experiences stress and DNA damage, some cells enter a state of permanent arrest where they refuse to divide but also refuse to die. These senescent cells linger in your tissues and secrete a toxic cocktail of inflammatory molecules that degrade surrounding healthy cells. Scientists identified this phenomenon as the Senescence-Associated Secretory Phenotype, linking it directly to arthritis, atherosclerosis, and overall tissue degeneration. Trials with senolytic drugs—compounds designed to specifically hunt and clear these zombie cells—have successfully rejuvenated tissue function in animal models and are undergoing human trials. By clearing out this inflammatory cellular debris, your body can dramatically reduce systemic inflammation and maintain healthier organ function well into later life.
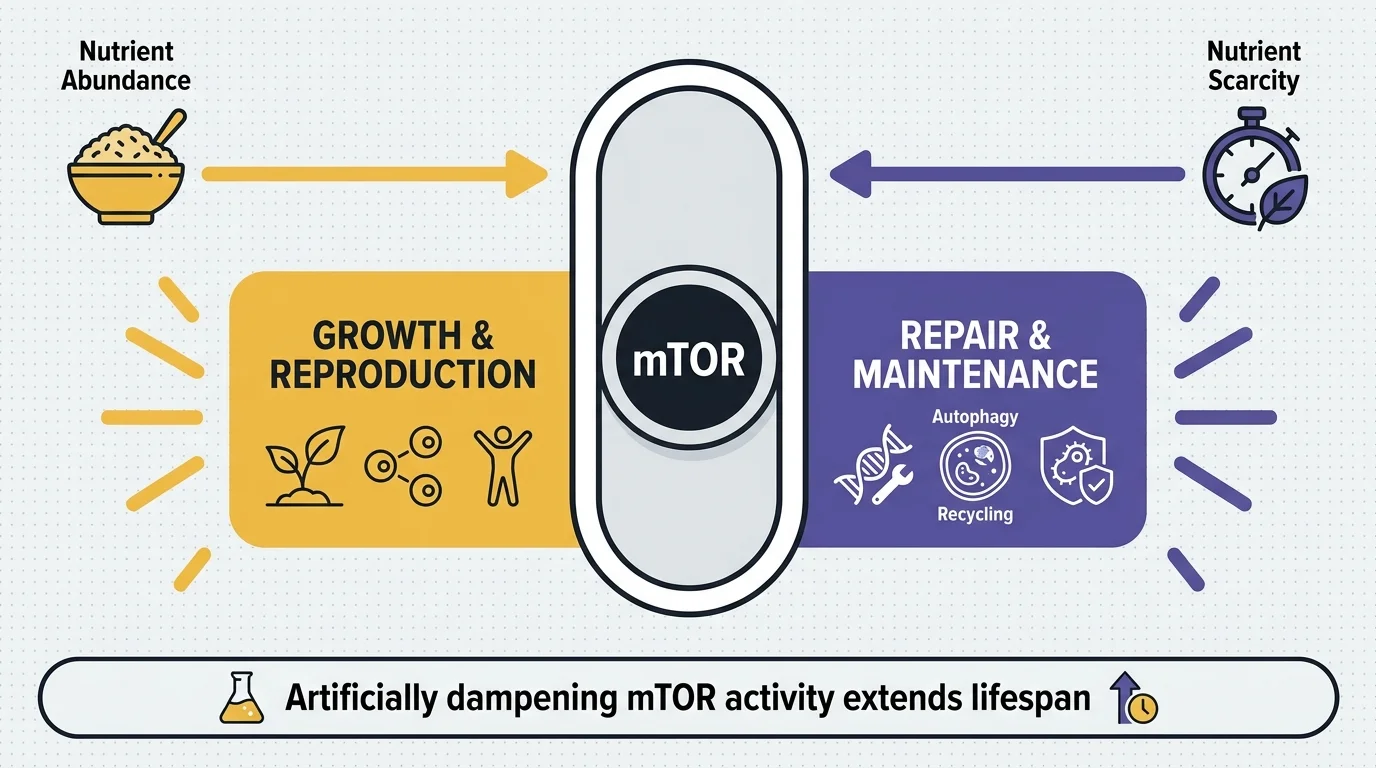
3. The mTOR Pathway and Nutrient Sensing
Hidden in the soil of Easter Island, scientists discovered rapamycin, a compound that unveiled one of the most critical aging pathways in biology: the mechanistic Target of Rapamycin. This protein acts as your cellular master switch for growth and reproduction. When food is abundant, mTOR drives cell growth; when nutrients are scarce, it dials back growth to focus on cellular repair and maintenance. Researchers found that artificially dampening mTOR activity significantly extends lifespan across multiple species, from yeast to mammals. This discovery explains why constant snacking and overeating accelerate aging, as they keep your growth pathways permanently switched on. Lowering your protein intake periodically or practicing intermittent fasting forces your cells into a protective maintenance mode.
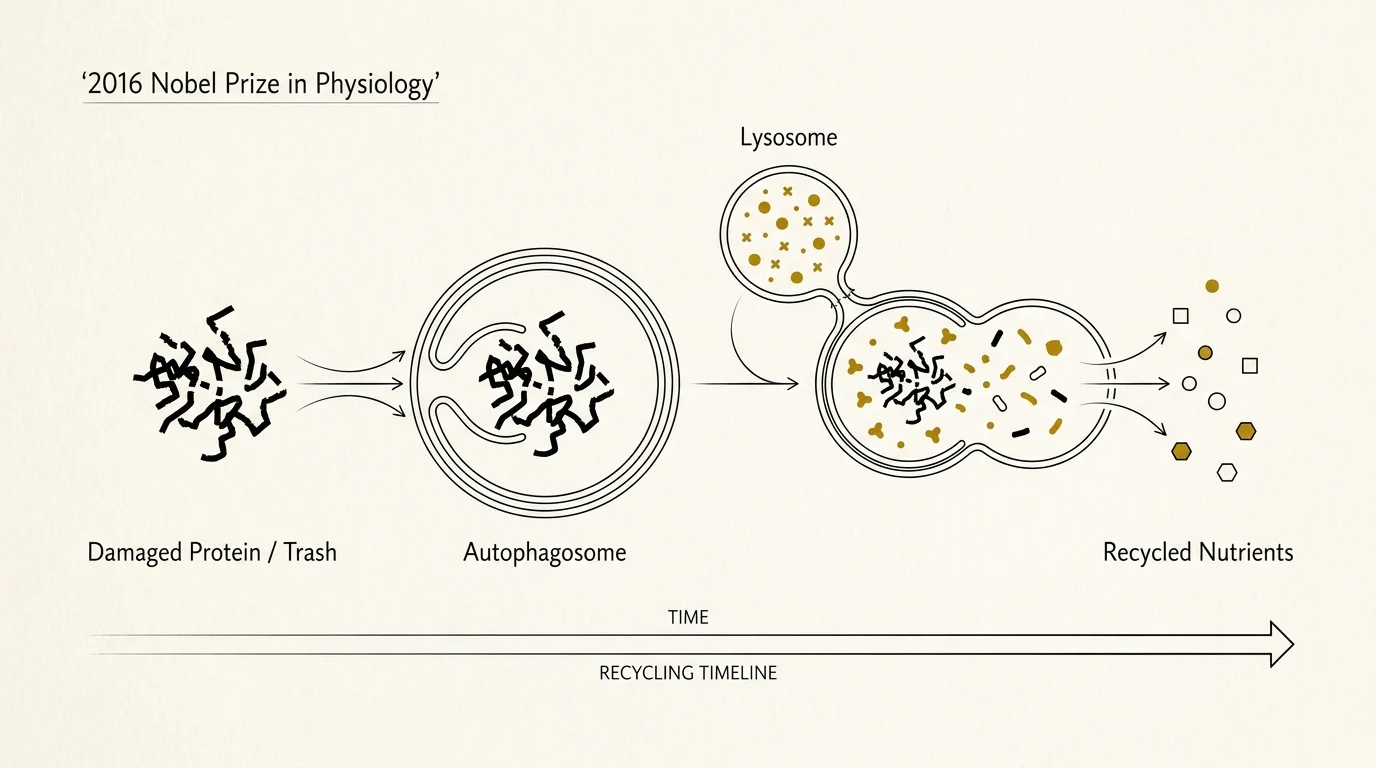
4. Autophagy and Cellular Recycling
Your cells act like tiny factories that inevitably accumulate damaged proteins and defective organelles over time. The discovery of autophagy, which won the 2016 Nobel Prize in Physiology, highlighted the intricate biological recycling system your cells use to encapsulate and break down this internal garbage. When autophagy slows down with age, cellular trash builds up and contributes to neurodegenerative conditions like Alzheimer’s and Parkinson’s disease. Fasting, vigorous exercise, and certain dietary compounds trigger this essential cleanup process. Understanding autophagy transformed how experts view age-related decline, shifting the focus toward therapies that artificially stimulate this recycling mechanism. By inducing autophagy through lifestyle interventions, you help your body clear out defective cellular machinery before it can trigger broader systemic failures.

5. Sirtuins and NAD+ Decline
Sirtuins are a family of proteins that serve as the emergency responders of your genome, rushing to repair broken DNA and regulate cellular health. However, these protective proteins require a coenzyme called NAD+ to function. Scientists made a crucial breakthrough when they observed that your NAD+ levels plummet by roughly fifty percent between youth and middle age, leaving your sirtuins without the fuel they need to fix daily genetic damage. This energy crisis causes your cells to lose their youthful identity and function. Clinical research now focuses on restoring these depleted levels using NAD+ precursors, which have shown immense promise in reversing vascular aging and restoring muscle endurance in aging models.
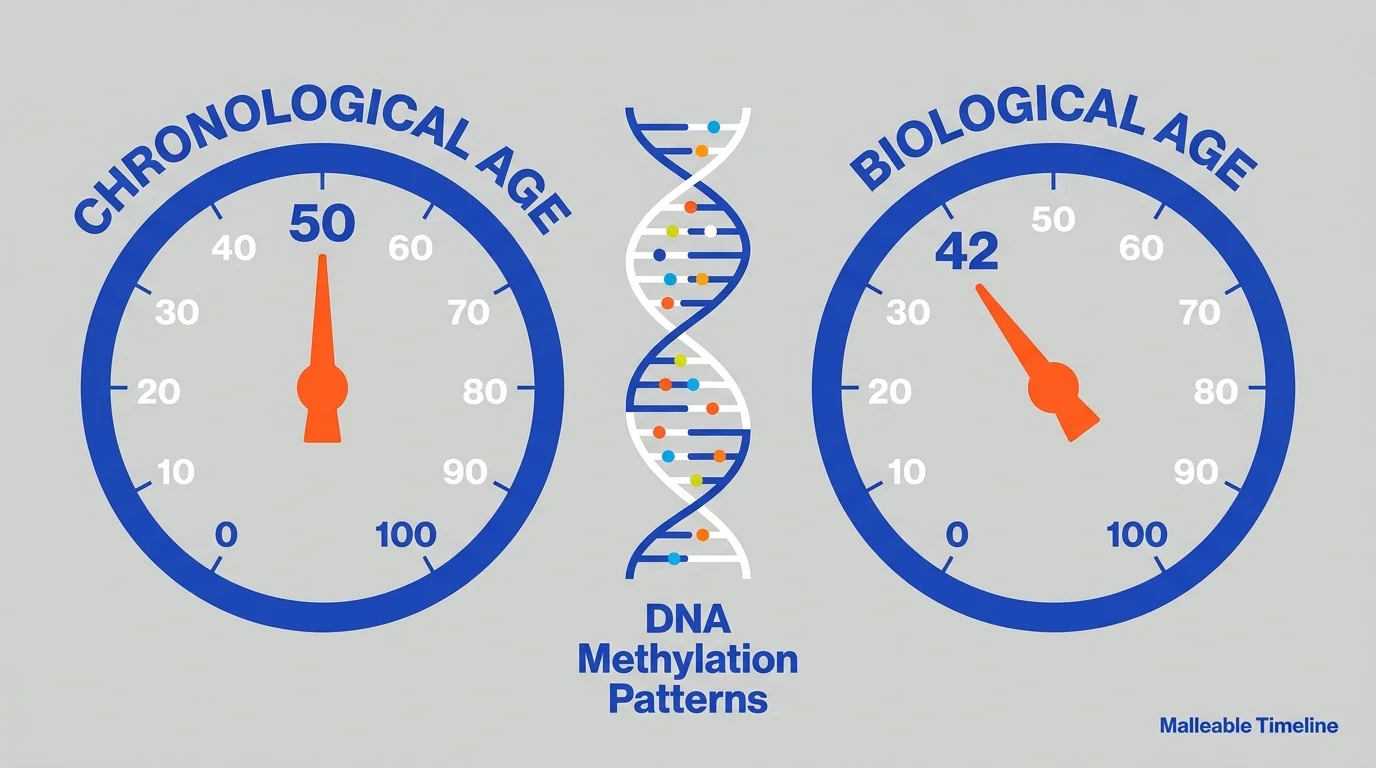
6. Epigenetic Clocks and True Biological Age
For decades, doctors relied entirely on your birth certificate to determine your age. That changed when researchers mapped DNA methylation—chemical tags that attach to your genome and turn specific genes on or off over time. By analyzing these predictable methylation patterns, scientists developed epigenetic clocks that measure your true biological age with astonishing accuracy. This discovery revealed that your organs can age at vastly different rates depending on your lifestyle, sleep quality, and environmental exposures. You can now use epigenetic testing to measure exactly how your diet or fitness routine impacts your aging process in real-time. This metric shifted longevity science from a guessing game into a precise, quantifiable discipline.

7. Caloric Restriction and Metabolic Shifts
The observation that reducing calorie intake without causing malnutrition extends lifespan is one of the oldest robust findings in aging research. However, modern science finally uncovered the mechanics behind this phenomenon. Data from the CALERIE trials—the first comprehensive human study on long-term calorie reduction—demonstrated that eating just fifteen percent fewer calories drastically lowers core body temperature and slows your basal metabolic rate, reducing oxidative stress. This metabolic shift forces your body to prioritize DNA repair over rapid cellular turnover. While extreme caloric restriction is difficult to maintain, these findings spurred the development of fasting-mimicking diets that trick your body into reaping the longevity benefits without the constant burden of starvation.

8. Yamanaka Factors and Cellular Reprogramming
In a breakthrough that sounds like pure science fiction, researchers discovered four specific proteins—now called Yamanaka factors—capable of wiping a cell’s developmental memory clean. By introducing these factors, scientists successfully reverted adult skin cells back to an embryonic stem cell state. This proved that aging is not a one-way street; the cellular software can be rebooted. Subsequent studies applied these factors in carefully controlled bursts to living mice, successfully rejuvenating their optic nerves and restoring lost vision without causing tumors. This partial cellular reprogramming represents the frontier of aging science, offering a future where doctors might systematically roll back the biological clock in failing human organs to restore their youthful function.

9. The Microbiome Connection to Longevity
Your gut houses trillions of bacteria that actively communicate with your immune system and brain, heavily influencing how you age. Researchers studying centenarians in specific blue zones discovered that individuals living past one hundred possess highly distinct and diverse microbial profiles compared to the average elderly population. These exceptional microbiomes produce specific short-chain fatty acids that suppress systemic inflammation and protect against harmful pathogens. As you age, your gut diversity typically collapses, leading to a leaky gut lining and chronic low-grade inflammation throughout your body. Maintaining microbial diversity through a fiber-rich, plant-heavy diet acts as a direct intervention against this age-related decline, proving that longevity is partly dictated by the microbes living inside you.
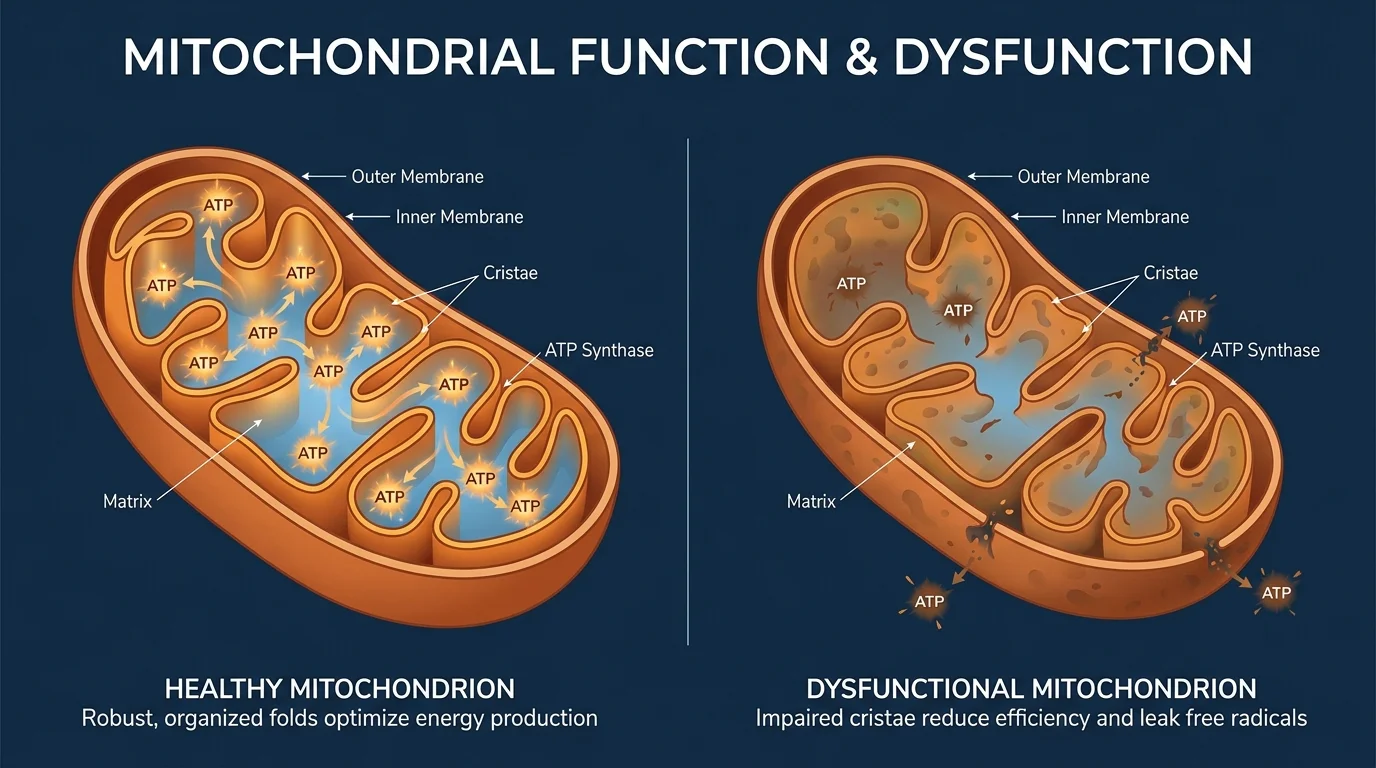
10. Mitochondrial Dysfunction and Energy Loss
Mitochondria serve as the microscopic power plants generating energy within your cells. Researchers identified that as these structures age, they become inefficient and leak highly reactive oxygen species—free radicals that damage surrounding cellular structures. This mitochondrial dysfunction explains the profound physical fatigue and muscle loss you experience as you get older. Scientists recently mapped how this energy deficit triggers a cascade of failures across various organ systems, particularly in energy-hungry tissues like the heart and brain. Modern interventions now target mitochondrial health through specific exercise protocols like high-intensity interval training, which forces your cells to destroy weak mitochondria and generate fresh, highly efficient power plants to sustain your energy levels.

The Global Impact of Extended Healthspan
Extending human life is not just a biological triumph; it carries profound implications for our global infrastructure. As international life expectancy continues to climb, healthcare systems face immense pressure to pivot from treating late-stage chronic diseases to prioritizing preventive longevity medicine. The World Health Organization projects that the proportion of the global population over sixty years old will double by 2050. If science successfully translates these aging discoveries into widely accessible therapies, societies could experience a massive demographic dividend. An extended healthspan means you can remain active, economically productive, and socially engaged for decades longer than previous generations. This shift reduces the crushing financial burden of age-related care and allows nations to harness the expertise of older populations. However, this progress also raises urgent questions about resource distribution and ensuring these groundbreaking therapies do not become a luxury reserved only for the wealthy.

Expert Voices on the Future of Aging
Leading gerontologists and longevity researchers agree that we are standing at an inflection point in human biology. Clinical researchers emphasize that the immediate goal is not necessarily radical life extension, but compressing morbidity—shrinking the amount of time you spend sick at the end of your life. Visionary researchers have spearheaded pushes for new clinical trials designed to prove to regulatory agencies that aging itself can be treated as a preventable medical indication. Policy makers and bioethicists caution that the societal rollout of longevity drugs must focus heavily on global equity. As scientists at the National Institute on Aging accelerate their funding for translational geroscience, the consensus remains unequivocally clear: lifestyle interventions remain your best defense today, while the pharmaceutical breakthroughs of tomorrow are systematically validated in clinical trials.
Frequently Asked Questions
Can I reverse my biological age right now?
While you cannot permanently stop the clock, you can definitively slow its ticking and even reverse certain epigenetic markers. Adopting robust exercise routines, optimizing your sleep architecture, and consuming a nutrient-dense diet actively improves your cellular health and lowers your measurable biological age. Doctors consistently note that your daily environmental exposures dictate your genetic expression far more than your birth year does.
Are longevity supplements actually effective?
The consumer supplement industry moves much faster than rigorous clinical science. While compounds like NAD+ precursors and certain senolytics show massive potential in animal models, human trials are still ongoing to determine optimal dosing and long-term safety profiles. You should approach supplements with a highly critical eye, prioritize lifestyle foundations first, and consult your physician before adding unregulated compounds to your daily regimen.
How does fasting influence the aging process?
Fasting acts as a beneficial environmental stressor that flips your metabolic switches from aggressive growth to conservative repair. By temporarily depriving your cells of constant nutrients, you naturally trigger autophagy to clear out cellular waste and lower mTOR activity. Both of these biological responses are strongly correlated with delayed tissue aging and improved metabolic flexibility.
Will scientists eventually cure aging entirely?
Most mainstream researchers view aging as an extremely complex, multi-system breakdown rather than a single disease with a singular cure. The realistic goal of modern science is to delay the onset of age-related diseases so effectively that you can live a vibrant, highly functional life right up until your final days. Immortality remains fiction, but extreme healthspan extension is rapidly becoming a biological reality.
Forward Look: What Comes Next
The next decade of longevity research will transition from mouse models into large-scale human applications. Artificial intelligence and machine learning now allow researchers to screen millions of molecular compounds in days, searching for the next generation of senolytics and metabolic regulators. Recent reviews in major scientific journals indicate that combined therapies—targeting multiple hallmarks of aging simultaneously—will likely be the key to significant breakthroughs. Clinical trials are already testing how combining cellular reprogramming with targeted metabolic drugs might synergistically repair organ damage. While the fountain of youth remains a myth, the blueprint for a longer, healthier human life is currently being written in laboratories around the world. Your daily choices dictate your baseline trajectory, but science is rapidly building the tools to help you exceed your biological limits.














