Unplanned moments often drive the greatest scientific breakthroughs, proving that a sharp observational eye matters just as much as a rigorous methodology. You might assume that world-changing research requires rigid planning and perfect execution, but serendipity repeatedly alters the course of human history. When a contaminated petri dish or a dropped flask interrupts standard procedures, attentive researchers pivot to investigate the anomaly rather than throwing the mistake away. This flexibility transforms laboratory blunders into life-saving medicines, revolutionary materials, and profound astronomical insights. By exploring these accidental discoveries, you gain a deeper understanding of how curiosity and unexpected errors combine to push the boundaries of technology, health, and our fundamental grasp of the universe.

Discovery 1: Penicillin and the Dawn of Antibiotics
In 1928, Scottish bacteriologist Alexander Fleming returned from a vacation to find a neglected petri dish covered in mold. Rather than tossing the ruined experiment, he paused. He observed that the invading mold—a strain called Penicillium notatum—actively halted the growth of surrounding Staphylococcus bacteria. This observation birthed the modern era of antibiotics. You see the profound impact of this biological accident in every hospital; infections that once carried a definitive death sentence became easily treatable. Today, health organizations track the evolutionary response of these pathogens closely, as researchers publish global assessments on antimicrobial resistance to prevent bacteria from outpacing our arsenals. The primary lesson extends beyond biology: when your intended process fails, examine the results before discarding the evidence.
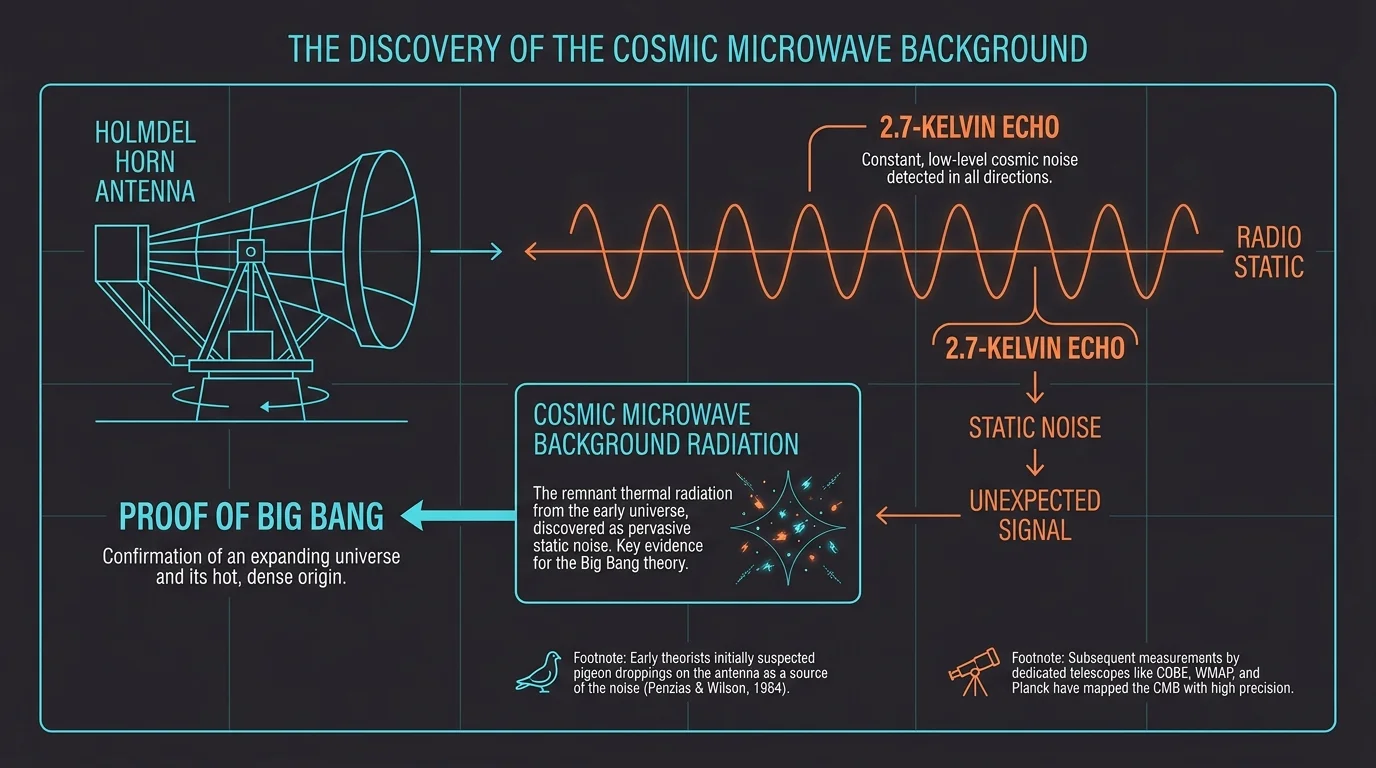
Discovery 2: The Cosmic Microwave Background Radiation
Astrophysicists Arno Penzias and Robert Wilson were not looking for the origins of the universe in 1964. They simply wanted to calibrate a massive horn antenna at Bell Labs to map radio signals emitted by the Milky Way. Instead, they encountered a persistent, uniform hiss that they could not eliminate—even after clearing out pigeons nesting inside the equipment. That stubborn static turned out to be the cosmic microwave background radiation. This 2.7-Kelvin echo of the Big Bang provided the concrete evidence required to confirm the universe’s explosive origin. You rely on invisible frequencies daily through telecommunications, yet this specific calibration error revolutionized cosmology. Their meticulous documentation continues to anchor modern astrophysics research archives globally.
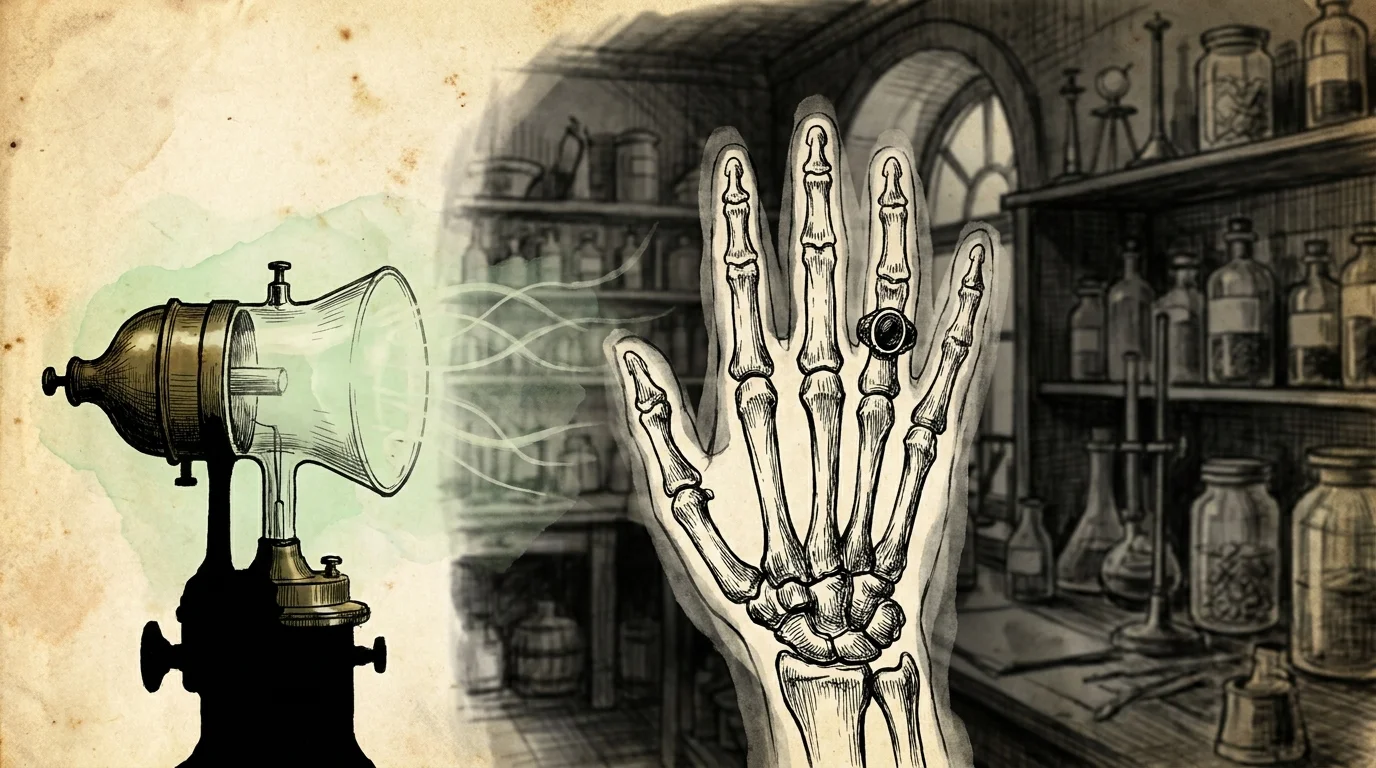
Discovery 3: X-Rays Unveiling the Unseen
While experimenting with high-voltage electrical currents inside a cathode-ray tube in 1895, physicist Wilhelm Conrad Röntgen noticed a faint glow emanating from a screen across his darkened laboratory. The tube was enclosed in thick black cardboard, proving that a new, invisible radiation was escaping. When he placed his wife’s hand in the path of these unknown rays, he saw the unmistakable shadows of her bones and wedding ring. Medical imaging sprang into existence virtually overnight. Modern healthcare facilities rely heavily on advanced variations of this technology to diagnose your internal injuries safely. Researchers continually refine protocols, documented in comprehensive health technology overviews, to minimize your radiation exposure while maximizing diagnostic clarity.
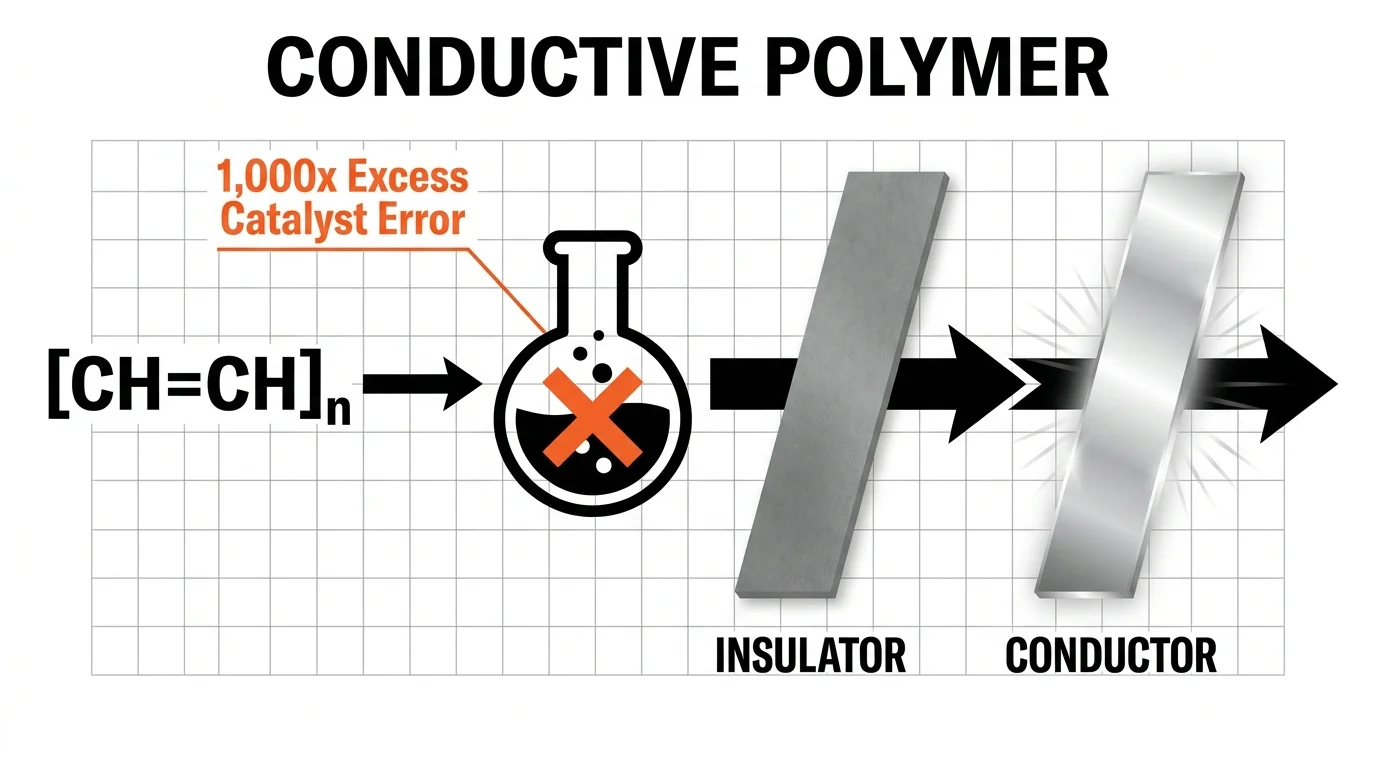
Discovery 4: Conductive Polymers from a Typo
You reasonably expect plastics to insulate against electricity, not conduct it. However, during the early 1970s, a visiting researcher in Hideki Shirakawa’s laboratory misread the chemical recipe for synthesizing polyacetylene, adding a thousand times the required catalyst. Instead of producing a dull powder, the reaction yielded a highly reflective, silvery film. Intrigued by the metallic appearance, researchers tested the unusual material and discovered that it could actively conduct an electrical current. This literal typo sparked the entire field of conductive polymers, eventually earning a Nobel Prize in Chemistry. Today, you carry the results of this accident in your pocket; the flexible touchscreens and light-emitting diodes that power your modern devices trace their lineage directly back to this specific laboratory blunder.

Discovery 5: The Implantable Pacemaker’s Wrong Resistor
In 1956, electrical engineer Wilson Greatbatch attempted to build an external oscillator designed to record rapid heartbeats. Reaching blindly into a box of spare components, he pulled out a 1-megaohm resistor instead of the standard 10,000-ohm resistor his blueprint required. Once he soldered the incorrect piece into place, the circuit failed to record sound; instead, it emitted electrical pulses in a rhythmic pattern that perfectly mimicked a healthy human heartbeat. Realizing the life-saving potential of this pulse, he shifted his focus to miniaturize the design, leading to the first successful implantable pacemaker. Millions of patients worldwide currently rely on this accidental innovation to maintain healthy cardiac function, proving that a simple component mismatch can dramatically extend human lifespans.
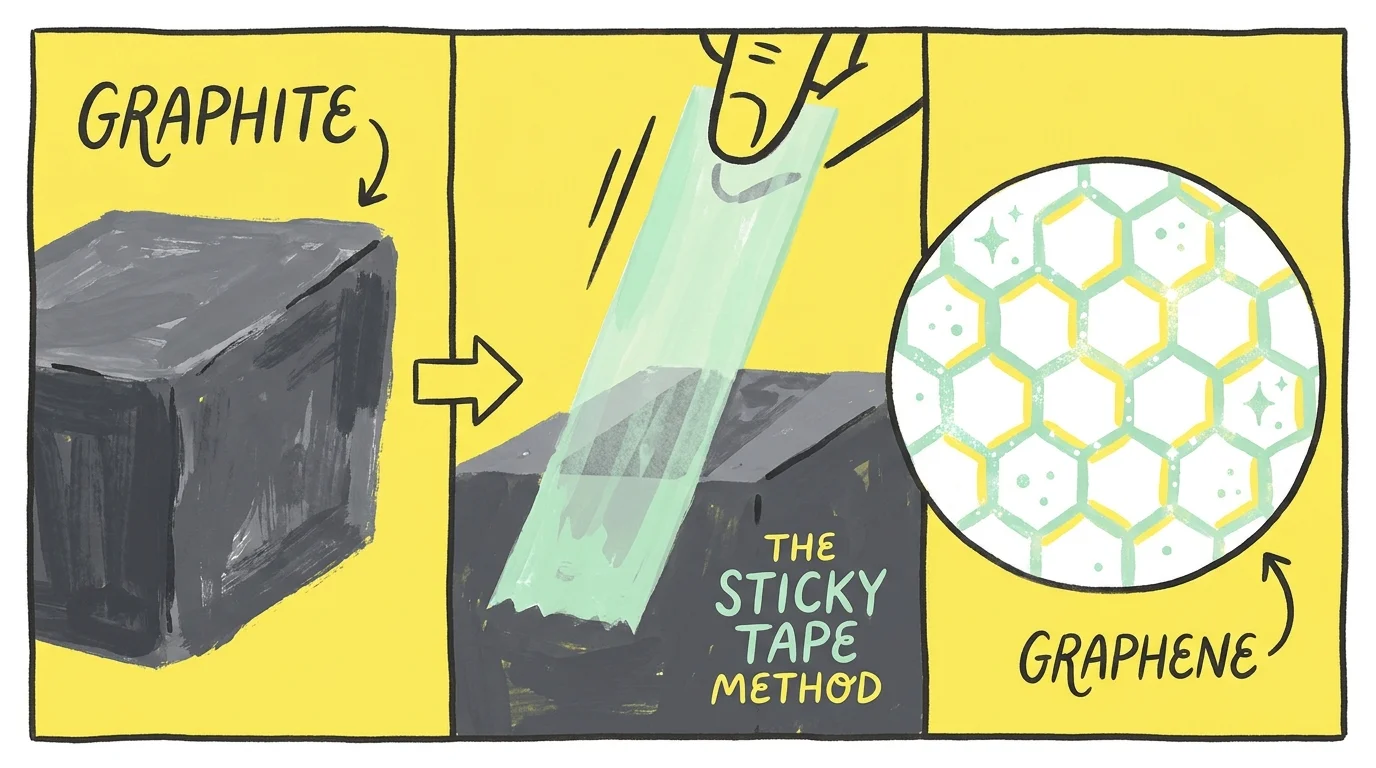
Discovery 6: Graphene via the Sticky Tape Method
At the University of Manchester in 2004, physicists Andre Geim and Konstantin Novoselov held routine Friday evening experimentation sessions, intentionally encouraging off-the-wall ideas. They used ordinary household adhesive tape to peel flakes of carbon from a block of graphite. By repeatedly applying and pulling off the tape, they progressively thinned the flakes until they isolated a flat layer of carbon exactly one atom thick. They had discovered graphene. This revolutionary material is incredibly lightweight, conducts electricity significantly better than copper, and is stronger than structural steel. You will soon see graphene transforming everyday commercial technology, from efficient electric vehicle batteries to advanced water filtration systems. This breakthrough, frequently highlighted in peer-reviewed materials science papers, demonstrates that playful curiosity yields serious scientific dividends.

Discovery 7: Radioactivity in a Dark Drawer
In 1896, French physicist Henri Becquerel planned to study phosphorescent materials by exposing uranium salts to sunlight, hoping they would emit energetic X-rays. Overcast Parisian skies thwarted his experiment, forcing him to stash the salts and heavily wrapped photographic plates together inside a dark drawer. Days later, he developed the unexposed plates anyway, fully expecting blank images. Instead, he found sharp silhouettes of the uranium crystals burned into the film. The material was actively emitting its own ambient energy without any external solar stimulation. This serendipitous drawer discovery opened the door to the entirely new field of nuclear physics, profoundly shaping modern society—from generating electricity at your local nuclear power plant to enabling targeted radiation therapies for cancer treatment.

Discovery 8: Safety Glass from a Clumsy Drop
Early automobiles featured dangerous plate glass windshields that caused severe lacerations during minor collisions. In 1903, French chemist Edouard Benedictus accidentally knocked a glass flask off his laboratory desk. The flask shattered violently, but remarkably, the jagged pieces stayed completely bonded together in their original shape. Benedictus discovered that the flask had previously contained cellulose nitrate, a liquid plastic that evaporated and left a strong, transparent, adhesive film coating the inside of the glass. He immediately realized the life-saving application for automobiles. Today, modern laminated safety glass protects you inside your car, your home, and your workplace. A simple clumsy moment fundamentally altered modern industrial manufacturing and drastically reduced global traffic fatalities.
Global Impact: How Accidents Shape Our Modern World
When you look across human progress, the global impact of accidental discoveries becomes impossible to ignore. These serendipitous events do not simply create isolated products; they launch entirely new scientific disciplines. The discovery of penicillin extended global life expectancy by decades, directly fueling population growth and economic expansion. Similarly, conductive polymers and graphene redefined materials science, allowing engineers to develop the lightweight technologies you depend on for daily communication. Furthermore, these unintentional breakthroughs highlight a crucial reality about scientific funding. Breakthroughs rarely adhere to a strict corporate timeline. Progress accelerates when societies support basic research that allows scientists the freedom to fail, tinker, and observe anomalies. This non-linear path from laboratory error to commercial application forms the backbone of modern innovation, reminding you that flexibility drives progress just as much as targeted invention.

Expert Voices: Cultivating an Environment for Serendipity
Louis Pasteur famously noted that chance favors only the prepared mind. Modern research directors actively echo this sentiment today, structuring their laboratories to capture the value of unexpected results rather than punishing them as failures. Laboratory managers increasingly encourage cross-disciplinary collaboration, knowing that a physicist might identify the value in a chemical error that a chemist might discard. Leading academic institutions host innovation forums where researchers present their failed experiments, explicitly searching for hidden value in anomalous data. Policy makers who draft sociological analyses of scientific progress frequently advocate for flexible grant funding. When scientists receive financial backing that allows them to pivot their methodologies mid-experiment, they can chase the unexpected phenomena that actually change the world. By intentionally engineering environments that tolerate safe mistakes, modern research institutions ensure that the next revolutionary material does not end up in the trash bin.
Frequently Asked Questions
Can scientists actively plan for accidental discoveries?
You cannot schedule serendipity, but scientists can systematically prepare for it. Researchers plan for accidents by building robust observation protocols and maintaining detailed laboratory notebooks. When an experiment goes off the rails, recorded data allows the scientist to retrace their steps and understand what variables caused the outcome. Preparation transforms a ruined experiment into a repeatable breakthrough.
How do modern funding agencies view serendipitous research?
Historically, grant agencies preferred rigid methodologies and predictable outcomes. However, scientific institutions increasingly offer flexible funding mechanisms. Organizations now recognize that strict adherence to a flawed hypothesis wastes resources, while giving brilliant minds the operational freedom to chase sudden anomalies frequently yields a much higher return on investment.
What role does modern technology play in finding accidents?
Technology acts as an amplifier for human observation. High-speed cameras, incredibly sensitive chemical sensors, and automated data logging catch fleeting anomalies that a human eye misses. By continuously monitoring the peripheral data of an experiment, modern technology captures the exact moment an accident happens, allowing researchers to study the event perfectly.
How can you apply this scientific mindset to your daily life?
You can harness the power of serendipity by shifting your perspective on failure. When a project at work or a recipe at home goes wrong, pause before starting over. Ask yourself why it failed and whether the unexpected result has an alternative use. Staying curious frequently leads you to creative solutions.
Forward Look: Preparing for the Next Happy Accident
The future of accidental discovery currently lies within the digital realm. As researchers feed colossal amounts of scientific data into advanced artificial intelligence networks, machine learning algorithms act as the ultimate prepared mind. These systems possess the processing power to sift through decades of complex chemical interactions and genetic sequences to highlight the anomalies human researchers overlooked. You will soon see breakthroughs emerge not from dropped flasks, but from algorithms flagging impossible data points that warrant real-world investigation. However, even with unprecedented computational power, the human element remains completely irreplaceable. An algorithm can spot a statistical deviation, but it takes human ingenuity to recognize how that anomaly might cure a disease or power a city. As we push deeper into an era of automated research, your curiosity and willingness to investigate the unexpected remain the true engines of global scientific discovery.