The Invisible Shield in Everyday Life
You probably know someone who walks through the height of flu season completely untouched. While colleagues succumb to congestion and fever, this person breathes freely, seemingly impervious to the microscopic threats swirling in our shared environments. You might chalk their resilience up to an impeccable diet, rigorous hand-washing, or simple genetic luck; however, modern biology reveals a far more intricate cellular choreography at play. Our immune system operates not merely as a defensive army, but as a highly perceptive ecosystem constantly negotiating with the surrounding world.
Understanding why certain people never get sick requires you to look beyond basic hygiene and examine the fascinating genetics, microbial partnerships, and evolutionary adaptations that shape human resilience. Researchers studying these exceptional individuals—often referred to as super-dodgers—are discovering that true immunity is an active, dynamic state of ecological balance. By analyzing the precise biological mechanisms that allow some humans to clear pathogens without experiencing a single symptom, scientists are radically rethinking our approach to medicine, disease prevention, and public health.
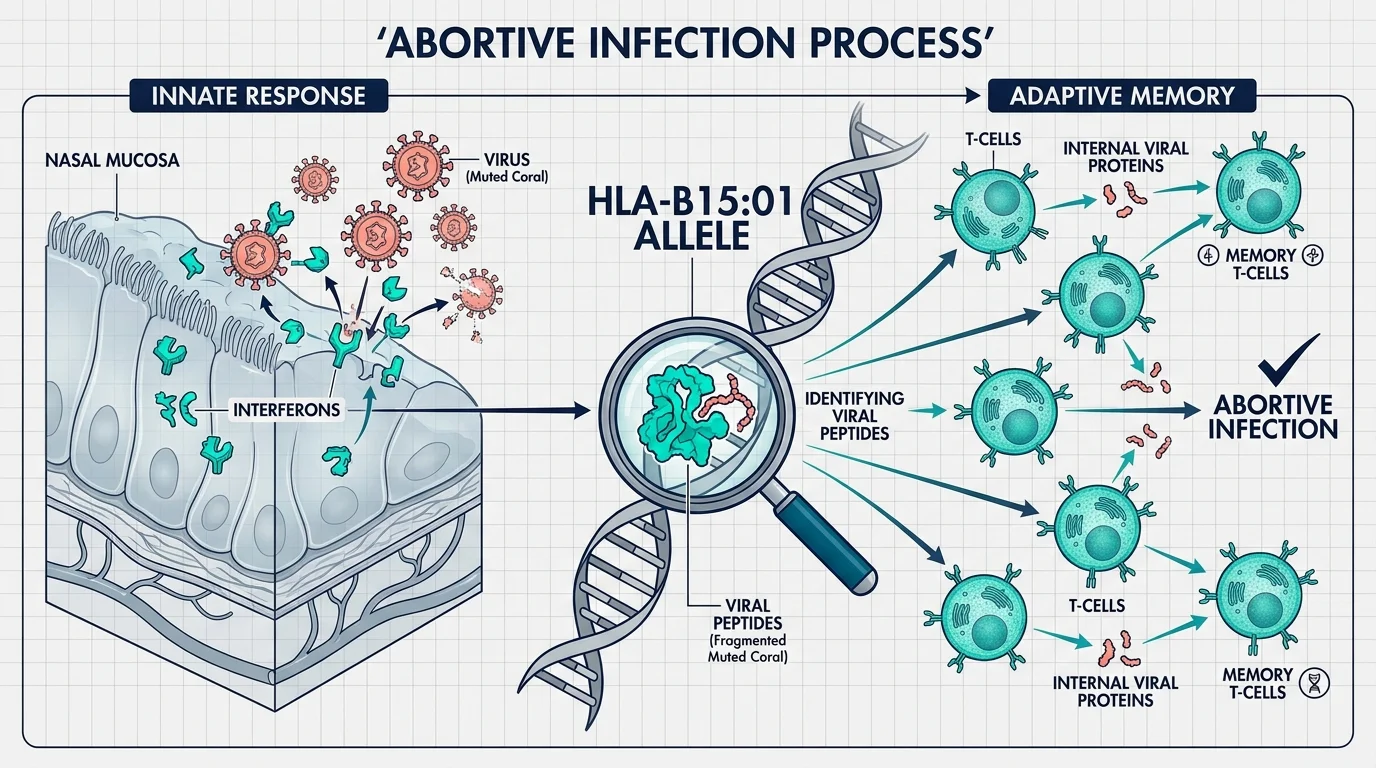
Decoding the Mechanisms of Abortive Infection
For decades, immunologists believed that exposure to a respiratory virus inevitably led to an infection, followed by an immune response that caused symptoms like coughing, inflammation, and fatigue. Yet, highly controlled experimental environments known as human challenge trials have turned this assumption upside down. In these studies, scientists deliberately expose healthy volunteers to precisely measured doses of pathogens, such as the influenza virus or rhinoviruses, and monitor their biological responses around the clock.
A landmark study published in the peer-reviewed journal Cell tracked healthy adults exposed to a respiratory virus. Surprisingly, a subset of these volunteers never tested positive for the virus and never developed antibodies, despite direct exposure. Researchers discovered that these individuals experienced what is known as an abortive infection. Their innate immune system—specifically, a rapid, highly localized release of signaling proteins called interferons in the nasal mucosa—shut down viral replication within hours. The virus was neutralized before it could establish a foothold, rendering the traditional antibody response entirely unnecessary.
Beyond the immediate innate response, your adaptive immune system plays a critical role through the deployment of memory T-cells. Unlike antibodies, which target specific proteins on the surface of a virus, T-cells can recognize internal viral proteins that remain consistent across different, but related, pathogen strains. If you have previously fought off a mild, endemic coronavirus—the kind that causes a common cold—your body might harbor cross-reactive T-cells. These experienced cellular sentinels quickly recognize and dismantle novel threats before you ever feel a tickle in your throat.
Genetics also dictate how efficiently your cells communicate during these microscopic encounters. The Human Leukocyte Antigen complex is a set of genes responsible for presenting fragments of foreign invaders to your immune system. Certain variants, such as the HLA-B15:01 allele, are incredibly efficient at showcasing viral peptides to T-cells. If you carry specific highly sensitive genetic variants, your immune system essentially operates with a high-definition radar, detecting and eliminating threats long before they multiply enough to trigger systemic inflammation.
Your Microbial Co-Pilots
Human cells only make up a fraction of your body; you are entirely dependent on the trillions of bacteria, fungi, and viruses living on your skin and within your digestive tract. You cannot fully understand human health without acknowledging these resident microbes. The gut microbiome acts as a fundamental training ground for your immune system. According to research highlighted by the Proceedings of the National Academy of Sciences, beneficial gut bacteria ferment dietary fibers to produce vital metabolites like short-chain fatty acids. These molecules circulate through your bloodstream, calming systemic inflammation and priming immune cells in your lungs—a connection scientists call the gut-lung axis.
People who rarely get sick often host a highly diverse microbiome. This internal biodiversity acts as an ecological buffer against invading pathogens. When your microbiome is robust, beneficial bacteria outcompete infectious invaders for resources and physical space along your mucosal linings. To actively support this internal network, you need to expose your system to a wide variety of plant fibers, fermented foods, and diverse natural environments. Every time you dig in an organic garden or hike through a dense forest, you breathe in environmental microbes that help calibrate your immune responses.

Ripple Effects: Reshaping Medicine and Planetary Health
The discovery of abortive infections and rapid T-cell clearance is revolutionizing how we design preventative medicine. Currently, most globally distributed vaccines are injected into a muscle. While intramuscular vaccines are extraordinarily effective at preventing severe disease and death, they primarily generate circulating antibodies in the blood. They are far less efficient at generating mucosal immunity in the nose and throat, which is exactly where respiratory viruses first invade.
By studying individuals who naturally stop infections at the point of entry, pharmaceutical researchers are aggressively developing next-generation mucosal vaccines, such as nasal sprays and inhaled aerosols. These experimental treatments aim to establish a permanent garrison of tissue-resident memory T-cells directly in your respiratory tract. If successful, these vaccines could theoretically block transmission entirely, creating an artificial version of the super-dodger phenomenon for the general population.
Furthermore, human immune resilience is deeply intertwined with broader ecological health. The biodiversity hypothesis suggests that contact with natural environments enriches the human microbiome, promoting immune balance and protecting against allergies and inflammatory disorders. Organizations dedicated to ecological conservation, such as the Earth Microbiome Project, emphasize that as we pave over natural habitats and rely heavily on industrial agriculture, we deplete the environmental microbes that have trained the human immune system for millennia. Protecting soil health and global biodiversity is not just an environmental crusade; it is a fundamental prerequisite for sustaining human immunity.

The Bioethics of Biological Privilege
While the science of natural immunity is awe-inspiring, it is heavily burdened by ethical and social complexities. Human challenge trials, which provide the clearest data on immune resilience, require volunteers to willfully expose themselves to potential harm. Institutional review boards must meticulously weigh the broader benefits to society against the localized risks to the individual, ensuring that volunteers are not economically coerced into participation and that emergency medical interventions are instantly available.
More pressingly, we must examine the concept of natural immunity through the lens of socioeconomic privilege. Chronic stress, sleep deprivation, and nutritional deficits profoundly degrade immune function by flooding the body with immunosuppressive hormones like cortisol. Individuals living in impoverished neighborhoods often face higher exposure to industrial pollution, limited access to diverse, fresh foods, and fewer green spaces. Consequently, their internal microbiomes and cellular defenses suffer.
When society praises individuals who never get sick, it often inadvertently ignores the socio-cultural determinants of health. Resilience is rarely just a matter of superior genetics or personal discipline; it is heavily subsidized by environmental safety, economic stability, and access to healthcare. Addressing global health disparities requires you to recognize that fortifying the human immune system demands systemic social reforms just as much as it demands advanced medical interventions.
Frequently Asked Questions
How do scientists safely conduct human challenge trials?
Conducting a human challenge trial involves extraordinary ethical and medical oversight. Researchers recruit healthy young adults who face statistically negligible risks of severe complications. The trials take place in specialized quarantine facilities with negative air pressure to prevent environmental leaks. Medical personnel monitor the volunteers continuously, taking daily blood and mucosal samples. If a participant’s symptoms cross a predefined threshold, researchers immediately administer antiviral rescue medications to neutralize the pathogen and halt the progression of the disease.
What are the risks of having an overactive immune system?
You might assume that a hyper-aggressive immune system is the ultimate goal, but true biological resilience relies on exquisite regulation. An overactive immune system that cannot accurately distinguish between dangerous pathogens and harmless environmental proteins leads to autoimmune diseases, such as lupus or rheumatoid arthritis, as well as severe allergies. People who never get sick do not possess aggressive immune systems; rather, they possess highly regulated, precise immune systems that know exactly when to attack and, equally importantly, exactly when to stand down.
Who funds the research behind natural immunity and resilience?
This complex research is funded through a mosaic of international partnerships. Government agencies, non-profit organizations, and prominent academic institutions pour billions of dollars into understanding immune resilience. Additionally, global consortiums dedicated to public health frequently collaborate with pharmaceutical companies to translate findings from basic biology into actionable therapeutics, such as new antiviral drugs and mucosal vaccines. Bioethics research regarding equitable access to these therapeutics is often supported by independent organizations like The Hastings Center.
Where is the field of immune resilience research heading next?
The frontier of immunology is moving away from generalized treatments toward personalized biological mapping. Scientists are focusing heavily on tissue-resident memory cells—the localized immune sentinels that live permanently in your skin, gut, and lungs. Future research will likely yield sophisticated immunotherapies that can be inhaled or swallowed to fortify specific vulnerable tissues. Furthermore, researchers are investigating how synthetic microbiomes could be engineered and prescribed to individuals whose natural microbial diversity has been compromised by repeated antibiotic use or environmental deprivation.
Observing the Invisible World
You are a walking, breathing ecosystem. Every time you navigate a crowded room or step into a dense forest, your immune system is actively reading the environment, learning from microscopic encounters, and recalibrating its defenses. The people who effortlessly dodge the seasonal flu are a testament to the astonishing capabilities of human biology—a synergy of favorable genetics, diverse microbial partnerships, and highly regulated cellular communication.
Understanding the science of health resilience changes how you view your body’s relationship with the natural world. Instead of viewing the environment merely as a source of disease, you can begin to see it as a vital partner in your survival. By deliberately engaging with biodiverse environments, prioritizing restorative sleep, and supporting conservation efforts that protect the microbial health of our planet, you actively participate in the stewardship of your own biology. Your health is not an isolated phenomenon; it is an enduring reflection of the thriving ecosystems both within you and around you.